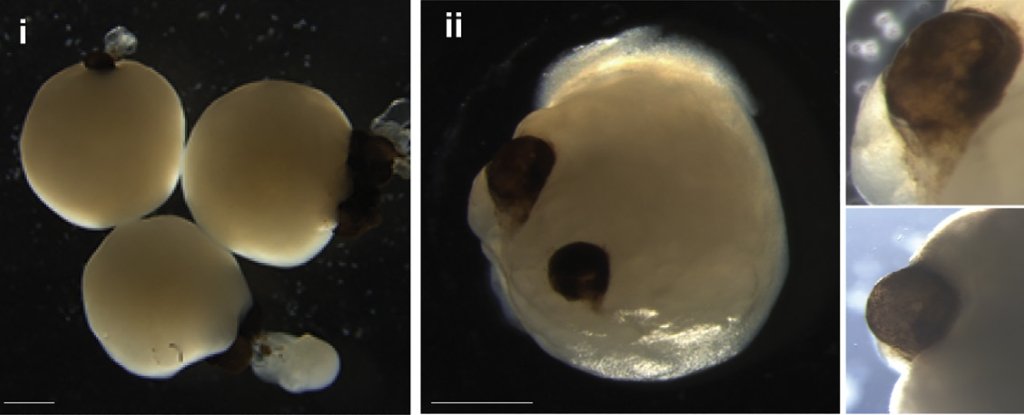
In a groundbreaking discovery reported in a 2021 paper, miniature brains cultivated in a laboratory from stem cells spontaneously evolved rudimentary eye structures, shedding light on the intricate process of eye differentiation and development. These tiny, human-derived brain organoids, resembling three-dimensional brain tissue blobs, showcased bilateral optic cups, mirroring the developmental patterns observed in human embryos. This astonishing revelation not only deepens our understanding of eye development but also holds significant potential for studying brain-eye interactions, modeling congenital retinal disorders, and advancing personalized drug testing and transplantation therapies.
Neuroscientist Jay Gopalakrishnan from the University Hospital Düsseldorf in Germany emphasized the remarkable capability of brain organoids to generate primitive sensory structures sensitive to light, housing cell types akin to those in the human body. Unlike true brains, these “mini brains” are structured from induced pluripotent stem cells, offering a unique platform for ethical research into drug responses, cell development, and, in this case, eye development.
The research team, led by Gopalakrishnan, departed from traditional methods of growing optic structures in isolation. Instead, they integrated optic cups into brain organoids to explore how these distinct tissues could coalesce and develop together. This innovative approach provides valuable insights into the collaborative growth of brain and eye tissues.
The process involved carefully nurturing mini brains with added retinol acetate in the culture medium to facilitate eye development. Astonishingly, the optic cups formed as early as 30 days into development, with clear visibility at 50 days, aligning with the timeline of eye development in human embryos. These optic cups harbored diverse retinal cell types, forming neural networks responsive to light and even incorporating lens and corneal tissue. The structures demonstrated retinal connectivity to regions of the brain tissue, a phenomenon previously unseen in in vitro systems.
Gopalakrishnan highlighted the reproducibility of the results, with 73 percent of the 314 brain organoids developing optic cups. The team envisions extending the viability of these structures for more in-depth research, opening avenues for personalized organoid and retinal pigment epithelial sheet generation for transplantation purposes. The development of these next-generation organoids holds promise in modeling retinopathies emerging from early neurodevelopmental disorders, marking a significant leap forward in the realm of regenerative medicine and neuroscience.
By Impact Lab