A diagram showing the team’s 3D bioprinted muscle-tendon up-close.
By PAUL HANAPHY
Researchers at Harvard Medical School and Sichuan University have developed a novel means of 3D bioprinting live human muscle-tendon tissues.
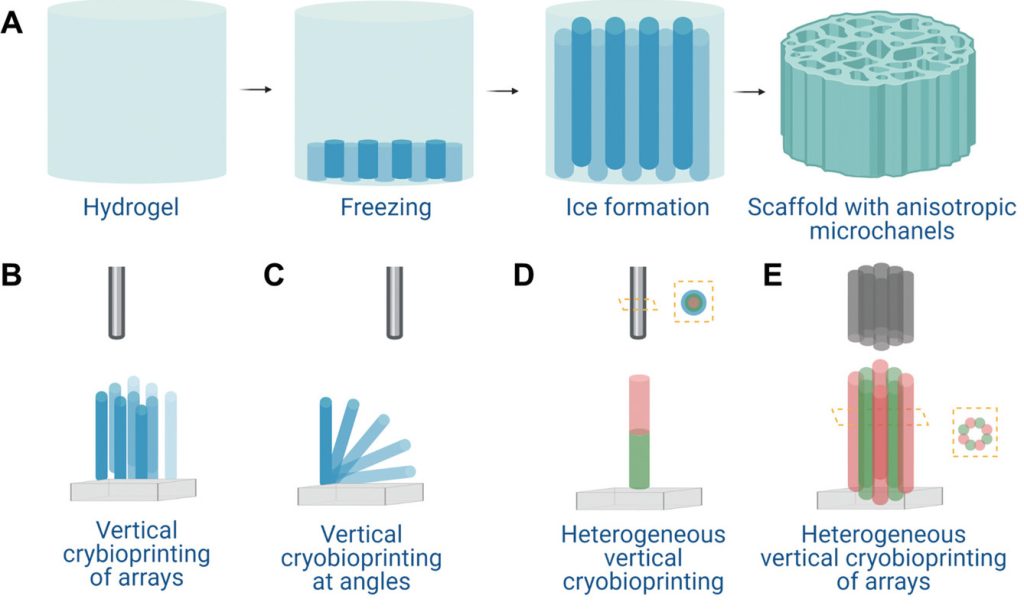
As opposed to normal extrusion bioprinting, which involves depositing cells along X and Y axes, the team’s ‘cryo-bioprinting’ process sees them frozen and stacked vertically, in a way that allows for the creation of freestanding, mixed-cell tissues.
According to the scientists, their technique also yields tissues that are more robust and versatile than those produced via conventional bioprinting, particularly when it comes to those anisotropic in nature, thus they say it could now find regenerative medicine, drug discovery, or personalized therapeutic applications.
An extrusion bioprinting alternative?
Few 3D bioprinting technologies have so far made it to market, and the field remains at a relatively early stage of development in this respect. However, of those used at an experimental level, the scientists say that many still rely on extrusion-based approaches, which are often functional, but when it comes to printing tissues that rely heavily on alignment, like muscle and nerve fibers, they tend to come up short.
To overcome these tissue-stacking issues, the researchers have therefore turned to ‘ice-templating,’ a freezing process that causes microchannels to form within cell-laden hydrogel-based structures once they thaw. Naturally, doing so would ordinarily damage the viability of such cells, so to prevent this, the team loaded theirs with the cryoprotective agents (CPAs) melezitose and dimethyl sulfoxide.
Once frozen, the researchers then used ultraviolet (UV) light to vertically cross-link this novel bio-ink, and extrude it into tissues composed of high-resolution, honeycomb-like microchannel networks, capable of supporting various different types of cell, whether they be skeletal muscle myoblasts or human umbilical vein endothelial cells.
“Our results indicate that [our] bio-ink, consisting of gelatin methacryloyl and CPAs, could be effectively used in vertical 3D cryo-bioprinting to enable cell encapsulation at high viability,” explained the team in their paper. “With the help of the interconnected, anisotropic, gradient microchannels formed by directional freezing during the process, the desired cellular alignments were also realized.”
‘Cryo-bioprinting’ a human tendon
Having demonstrated the viability of their approach, the scientists went on to assess its efficacy in the creation of more complex multiple cell-type tissue structures. To achieve this, the team initially used a 3D bioprinter armed with coaxial nozzles to deposit cells into vertical core-shell structures, in a process that allowed them to precisely control material deposition, while using up to eight different inks.
Critically, within these tissues, the researchers also found that their approach allowed them to create customizable, anisotropic microchannels that facilitated cellular growth, albeit with varying levels of success. After several trial prints, the bio-ink which combined melezitose and DMSO of 8% and 10% respectively proved to be the most effective, yielding tissues that fluctuated from 60-80% cell viability.
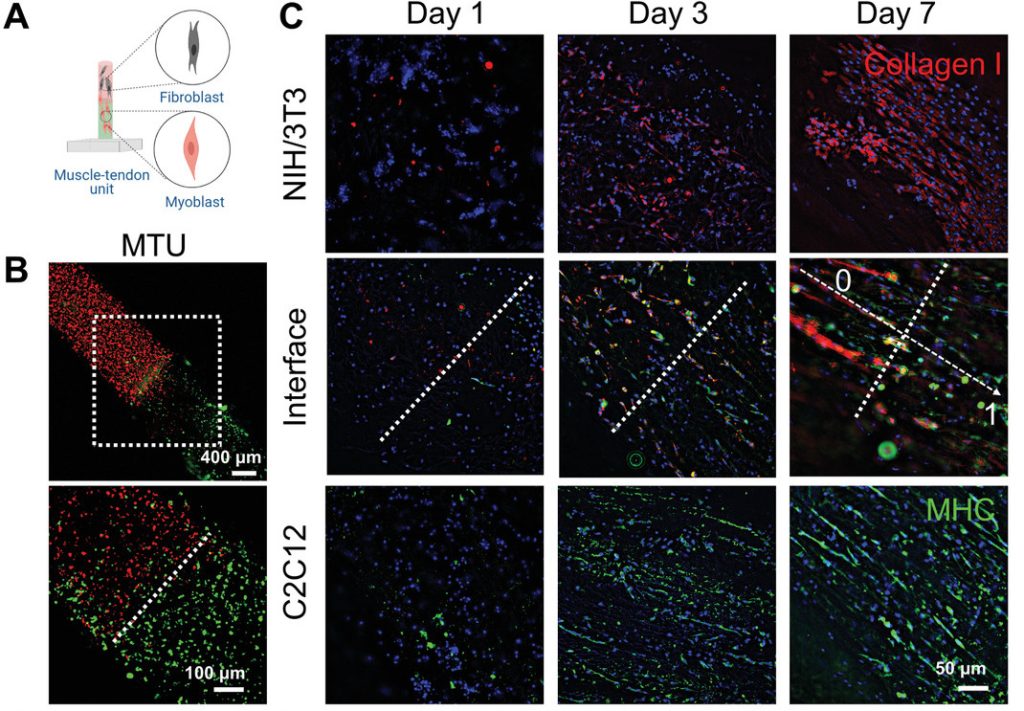
However, despite these mixed results, which the scientists attributed to potential variations in their experimental processes, they went on to take their approach a step further, by applying it in the creation of a tendon ‘junction,’ a tissue mass that in its organic form, transmits the force of a muscle’s contraction through the tendon to the human skeletal system.
During this process, cryo-bioprinting was said to prove ideal, as it enabled the creation of a junction in which its lower cells were highly aligned, with its fibroblasts less so, mimicking the structure of its natural counterpart. Once printed, the resulting tendon was cultured for seven days, in which its muscular and vascular bio-inks demonstrably interfaced, growing into a dense microvascular network.
As a result, even though the team admits their method currently has print height limitations which prevent its use within certain in-vivo applications, they maintain that it could yet be deployed in the creation of musculoskeletal models, with the potential to help develop patient-specific treatments and advance our understanding of the human body.
“It is rationally anticipated that our vertical 3D cryo-bioprinting strategy may find broad usage in engineering a variety of tissues that feature-oriented internal cellular and extracellular matrix arrangements,” concluded the team in their paper. “Another possible utility of the method lies in the creation of in vitro musculoskeletal models for biological studies, drug discovery and personalized medicine.”
Novel approaches to bioprinting
Given that 3D bioprinting is an emerging technology, it’s hardly surprising that its format is continually subject to change, with researchers constantly bringing innovative new ideas to the field. Just last month, scientists at the UK’s University of Birmingham and University of Huddersfield, revealed that they had developed a novel skin 3D bioprinting technique that enables the treatment of chronic wounds.
Elsewhere, on a more commercial level, Inventia Life Science raised $25 million towards the development of its RASTRUM 3D bioprinting technology in December 2021. In effect, the firm’s approach is designed to enable the layering of cell-loaded droplets onto one another at pace, in a way that allows them to join on contact and doesn’t affect their overall viability.
Looking even further back, researchers at Imperial College London have also experimented with cell-freezing as a means of bioprinting viable human implants. In a paper, now published four years ago, a team there sought to combine 3D printing and cryogenics as a means of replicating the texture of soft tissues in the body, and fooling the brain and lungs into accepting grafts as if they were organic.
The researchers’ findings are detailed in their paper titled “Vertical Extrusion Cryo(bio)printing for Anisotropic Tissue Manufacturing.”
The study was co-authored by Zeyu Luo, Guosheng Tang, Hossein Ravanbakhsh, Wanlu Li, Mian Wang, Xiao Kuang, Carlos Ezio Garcia mendez-Mijares, Liming Lian, Sili Yi, Junlong Liao, Maobin Xie, Jie Guo, Zongke Zhou and Yu Shrike Zhang.


